JeffW
#1130
My age range is 55 to 60.
adssx
#1131
Prediction of Cumulative Exposure to Atherogenic Lipids During Early Adulthood 2024
Just published. Very interesting:
We included CARDIA (Coronary Artery Risk Development in Young Adults Study) participants who were free of cardiovascular disease before age 40 years, were not taking lipid-lowering medications, and had ≥3 measurements of LDL-C and non–HDL-C before age 40 years. First, we assessed the ability of a 1-time measurement of LDL-C or non–HDL-C obtained between age 18 and 30 years to predict the quartile of cumulative lipid exposure from ages 18 to 40 years. Second, we assessed the associations between quartiles of cumulative lipid exposure from ages 18 to 40 years with ASCVD events (fatal and nonfatal myocardial infarction and stroke) after age 40 years.
The absolute values of non–HDL-C and LDL-C that predicted membership in the top quartiles with the highest simultaneous sensitivity and specificity (highest Youden’s Index) were >135 mg/dL for non–HDL-C and >118 mg/dL for LDL-C; the values that predicted membership in the bottom quartiles were <107 mg/dL for non–HDL-C and <96 mg/dL for LDL-C. Individuals in the top quartile of non–HDL-C and LDL-C exposure had demographic-adjusted HRs of 4.6 (95% CI: 2.84-7.29) and 4.0 (95% CI: 2.50-6.33) for ASCVD events after age 40 years, respectively, when compared with each bottom quartile.
Single measures of non–HDL-C and LDL-C obtained between ages 18 and 30 years are highly predictive of cumulative exposure before age 40 years, which in turn strongly predicts later-life ASCVD events.
Massive HR. I guess it’s now clear that you want non-HDL-C/LDL-C/apoB always below 100 mg/dL during your lifetime and that it provides massive protection.
Question still open: is there any benefit for healthy young people to go from 100 mg/dL to 70 mg/dL or lower? (I think evidence points to “yes” but it’s not as clear as what we have above)
6 Likes
mccoy
#1132
We should run simulations on a model like Ference et al., described in another thread.
However, I couldn’t find the whole article yet and I have a few doubts on a sentence, they say the top and bottom quartiles, but maybe they mean the top and bottom quartile singular? Otherwhise we are talking above or below the median.
mccoy
#1133
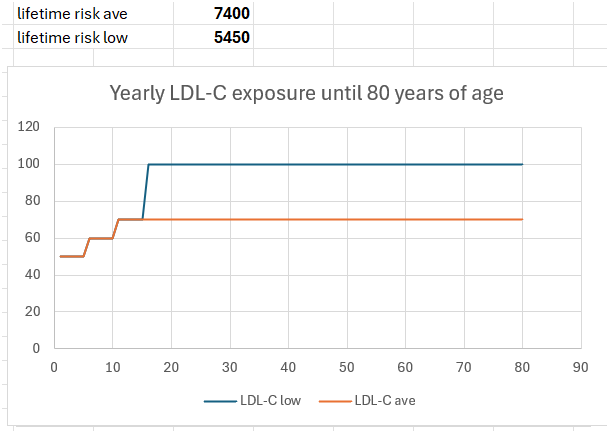
This is what may happen at 45 years of age, X-axis is age, Y-axis il LDL-C in mg/dL, red line is lower (70 mg/dL) exposure starting from 15, blue is higher starting from the same age (70 mg/dL). The lifetime cumulative exposure in the higher exposure is 900 (mg/dL)* years greater. both cumulative exposures, 3000 and 3900 are below the 5000 first threshold, 1% value of 5000, but the lower measurements carry a nontrivial advantage for later years. The model should be extended until 70-80 years to see what happens at a constant value. What happens according to Ference et al. is that the less exposed at 80 reach a cumulative risk of bout 1%, and the more exposed reach a cumulative risk of about 5%, probably a nontrivial difference.
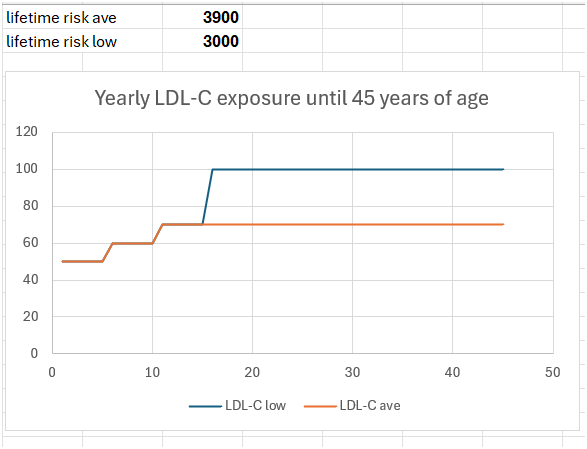
The values at 80 years would be 5450 and 7400 respectively
1 Like
Virilius
#1134
My criticism of this lifetime exposure model is that if it is indeed true, we wouldn’t expect statins to show an effect after just a couple years.
2 Likes
Davin8r
#1135
Anti-inflammatory effect along with less influx of new LDL particles = plaque stabilization/calcification?
2 Likes
Virilius
#1136
But you would have been still exposed to X amount of LDL over the past years so it shouldn’t matter according to the exposure model.
Davin8r
#1137
The exposure model says that statins don’t have an anti-inflammatory effect on plaques? I thought it just says that plaque burden is generally higher with longer term exposure to LDL-c over a lifetime. Both can be true.
1 Like
mccoy
#1138
My previous knowledge is that significant temporal horizons are 10 yrs (guidelines) and 30 years (Snidermann et al.).
The article reports what follows in regard to statins and shorter times of exposure. The statement is not very coherent with the graph exhibited in the article, where cumulative risk is a monotonic function, always increasing. There is actually an apparent contradiction.
Indeed, clear evidence exists demonstrating that the benefit
of lowering LDL-C levels increases with each additional year of LDL-C-
lowering. In randomized trials, reducing LDL-C levels by inhibiting
hydroxy-methylglutaryl CoA reductase using a statin or by inhibiting
proprotein convertase subtilisin–kexin type 9 (PCSK9) with a mono-
clonal antibody both reduced the risk of major cardiovascular events
by 10% in the first year of treatment and by 20% in the second year of
treatment such that, after 2 years of therapy, both statins and PCSK9
inhibitors reduced the risk of major cardiovascular events by 15% per
millimole per litre reduction in LDL-C levels12,13,81–84. In the statin tri-
als, this benefit increased to 18% per millimole per litre reduction in
LDL-C levels after 3 years of therapy, 20% after 4 years and 22% after
5 years12,13,82–86. Furthermore, there is compelling evidence that the
clinical benefit of lowering LDL-C levels continues to increase with
each additional year of treatment well beyond the duration of LDL-C
lowering observed in the randomized trials.
3 Likes
The issue is that the variables differ by individual, so such global averages must be seen in that light. For some, a given level of ApoB/LDL exposure over time might result in less/more plaque accumulation. So I’m not going to excitedly calculate my exposure according to these graphs. My CAC score at 65 is zero - are these formulas relevant to me?
Given such individual variability, doesn’t it make sense to have regular CAC score tests? As your age increases, the value of the test goes up. But for some unlucky folks who have more accumulation of plaque at lower lipid levels at a shorter time frame, a CAC score test at 35 might catch a problem earlier. So it seems advisable for everyone to get a CAC test early, like 30’s, to catch aggressive plaque development - a kind of screening tool.
But as a precautionary measure you obviously want to lower your ApoB levels as early as possible, at least to below LDL of 70 or so.
2 Likes
mccoy
#1140
The model presented reflects the average risk of a whole population, or cohort, subjected to a determined cumulative exposure to LDL-C . Sometimes these models may report the variability in response, with percentiles, quartiles and so on. An individual like you with a zero CAC at 65 may perhaps consider him/herself in the lower percentiles of risk, a low responder to LDL-C, as it were.
This inference is not possible in the Ference et al. model.
Of course the above assuming that CAC score at determined ages really reflects the state of responder or not responder to LDL-C
1 Like
Neo
#1141
@JeffW @forceofnature which lab are these with
I’m always below the threshold with Quest, but that threshold is higher
Anyone is US know how to measure even more sensitive than <0.15?
1 Like
Jonas
#1143
Average person don’t have access to IL-6 blockers, how about a low does intermittent prednisone which is used in conditions characterized by inflammation or immune system overactivity.
https://www.nature.com/articles/s41586-024-07282-7
1 Like
adssx
#1144
Cardiovascular benefits of statin plus ezetimibe combination therapy versus statin monotherapy in acute coronary syndrome: a meta-analysis of randomized controlled trials 2024
Six RCTs comprising 20 574 patients with ACS were included, of whom 10 259 (49.9%) were prescribed ezetimibe plus statin. The patient population had an average age of 63.8 years, and 75.1% were male. Compared with statin monotherapy, ezetimibe plus statin significantly reduced major adverse cardiovascular events (MACE) (risk ratio 0.93; 95% CI 0.90–0.97; P < 0.01) and nonfatal myocardial infarction (risk ratio 0.88; 95% CI 0.81–0.95; P < 0.01). There was no significant difference between groups for revascularization (risk ratio 0.94; 95% CI 0.90–1.00; P = 0.03), all-cause mortality (risk ratio 0.87; 95% CI 0.63–1.21; P = 0.42), or unstable angina (risk ratio 1.05; 95% CI 0.86–1.27; P = 0.64).
1 Like
Bempedoic acid is a relatively newish entrant into the ApoB/LDL lowering space. No question that BA lowers LDL. It also appears to lower MACE rates in statin intolerant subjects. Well, statin intolerant might be physiologically different from statin tolerant, so the question becomes does BA work equally well in statin tolerant to lower MACE rates.
More saliently, is BA equally effective as statins in lowering CV events per unit of lowered LDL.
It appears that perhaps yes:
Good authors and institutions, but suboptimal conflicts of interest.
2 Likes
adssx
#1146
BA seems good, but its safety profile is not as good as ezetimibe. And it doesn’t lower LDL much. If obicetrapib is approved, I can imagine BA disappearing from the picture. If not, I assume BA+EZE will be used way more often among statin-intolerant people.
1 Like
Good find. Slightly underwhelming results for the addition of ezetimibe, no? Or am I missing something?
adssx
#1148
I don’t know. What’s the MACE reduction for statin therapy in people with ACS? What about other therapies?
It’s also possible that there is one-third of high-respondents (hyperabsorbers) who benefit a lot from ezetimibe while the other two-thirds benefit less. (e.g. -15% MACE for some and -5% for the others giving an average of about -7%).
As to BA safety, compared to eze, debatable. But at least compared to statins doesn’t seem to raise diabetes risk:
1 Like