Jjazz
#1
Here is one likely risk of taking rapamycin doses too high - accelerated thymic involution:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9360838/
This is the process that the TRIM and TRIM-X trials succesfully reversed with HGH/DHEA/metformin. In this paper, 2.5 micrograms/mouse per day (5 days/week) for one month caused an approximately 50% reduction in thymus weight. Using allometric scaling for body surface area, this puts the human dose equivalent at about 14.125 mg/day for a 70kg individual, so much higher than doses typically used for longevity.
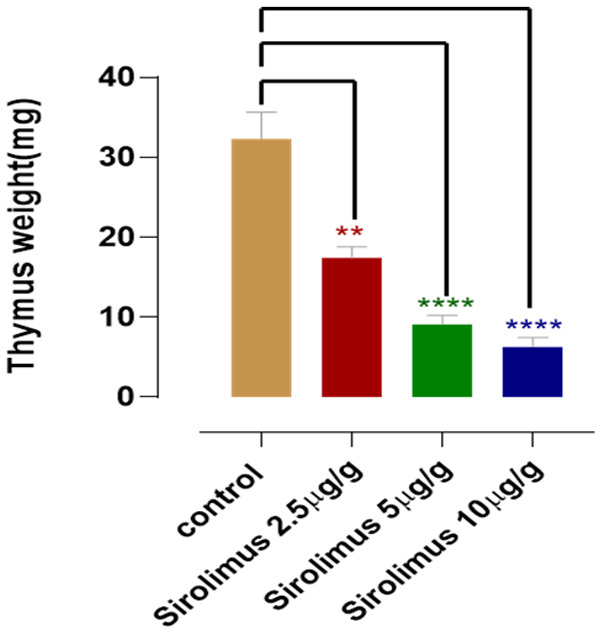
This is definitely something we want to avoid! I really hope the marmoset studies will perform histology on thymic tissue as well, along with looking at blood immune cell populations as a proxy for thymic function. It would be very reassuring if this wasn’t happening in primates.
15 Likes
Yes, but rapamycin extends health span and lifespan in all mammal studies I have seen.
It would be surprising if it doesn’t have the same effects on humans.
I will be coming up on my 3-year history of taking high-dose rapamycin with no ill effects noted. The slight rise in lipids and glucose was easily mediated by taking a few other probable life-extending medications.
10 Likes
Herm
#3
I hope they extend this to lower doses, 0.25, 0.5 and 1 microgram/g in a future study since 50% at 2.5 micrograms/g is a robust response.
2 Likes
I presume this is daily dosing and results from inhibition of cell division in part.
5 Likes
Jjazz
#5
I don’t think this calls the lifespan extension in animal studies into question, but it does suggest that there may be a dose ceiling wherein lifespan benefits will be offset by negative impacts on immune function from thymic involution.
What we really need is complete dose/response curves for each impact, showing the minimum effective dose (MED) and maximally effective dose for lifespan extension and the maximum tolerated dose (MTD) for avoiding thymocyte death and other toxicities. As of now, we still don’t really have any of these metrics from the mouse studies. Any new drug development program would work to find these in animal models before dosing in humans, but since rapa has long been approved, there is little incentive for a drug company to develop these data.
8 Likes
Karel1
#6
Mice received intraperitoneal injections of sirolimus (treated group) or Dimethyl sulfoxide (DMSO: control group) for 5 consecutive days each week for a total of 4 weeks.
We cannot calculate the equivalent dose of intraperitoneal injections to human oral dose as if it were oral and not intraperitoneal. Furthermore the amount of DMSO solvent differs between groups.
I would like to know if there is a function test available in humans and then compare if effective thymus “size” of an older long time user on rapa is reduced below that of an healthy young adult in the age group 20-30.
Jjazz
#8
@Karel1 , you are absolutely correct regarding ROA. The IP injections used here are 100% bioavailable, unlike oral dosing which is somewhere in the range of 10-20% bioavailable for rapamycin. As a rough approximation, you can multiply the dose here by 5 to get an “equivalent” oral dose. These calculations will be different for labs using nanoparticle (e.g., Eudagrit) formulations for oral rapamycin, since that is more bioavailable. Overall, the doses here are much higher than those typically used to study longevity in mice.
@DeStrider , this study did use DMSO vehicle injections in their control group, so I don’t think you can point to DMSO as the cause of the thymic degeneration. Unfortunately, the DMSO wasn’t perfectly controlled, with the low dose group receiving half of the amount of the control group, the mid dose group receiving the same DMSO as the control group, and the high dose group receiving twice the DMSO as the control group. In any case, rapamycin appears to be driving the effect.
1 Like
DMSO is, however, bioactive. Hence any variation in DMSO has to be recognised as a potential cause of issues.
Jjazz
#10
@Karel1, the easiest way to look at this in humans would be to measure thymic volume with MRI. This shows clearly the overall volume, and the volume occupied by fat (involuted) or active thymus tissue. As for a “functional” readout, circulating numbers of various immune cell populations are frequently used. Both of these types of measurements could be compared against ranges typically observed for various age groups.
1 Like
Jjazz
#11
Yes, but the control group received more DMSO than the low dose group, yet the low dose group ended up with half of the thymic volume of the control group. If DMSO was driving the effect, you would expect to see the opposite.
If you want a perfectly controlled comparison, pay attention only to the middle dose, since that group received the same amount of DMSO as the control group. Those animals lost >2/3 of their thymic tissue.
2 Likes
Jjazz
#12
This isn’t the first study to find that rapamycin causes thymic degeneration in mice. See here, as well:
MTOR signaling is essential for the development of thymic epithelial cells and the induction of central immune tolerance
“Pharmacological inhibition of MTOR activity by rapamycin (RPM) causes severe thymic atrophy and reduction of thymic epithelial cells.”
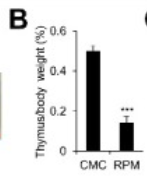
This is after only 7 days of 1.5 mg/kg IP dosing, with no DMSO 
1 Like
The problem is that there can be u shaped dose curves. Still we know rapamycin
inhibits cell division
Glad I’m doing my little TRIIM protocol along with the Rap program.
4 Likes
Karel1
#15
This is an extreme high dose.
2 Likes
Jjazz
#16
Actually, these doses are in the range of those used in the ITP studies for lifespan extension, even after you account for the differences in route of administration. Since 1) mice in these studies are living in relatively clean, low pathogen environments, and 2) circulating immune cells persist long after thymic degeneration takes place, it is possible that any early thymic involution wouldn’t impact their lifespan.
It seems to me that this is a very underappreciated potential side effect from rapa. We need to know if it still happens with intermittent dosing and at lower dose levels such as the ones used in humans. Since the impact of early thymic involution won’t manifest for many years, it is entirely possible that this is happening in humans at the current dose levels, but studies haven’t detected the impacts since they haven’t followed patients for long enough or looked at the right variables. Or maybe it isn’t happening at all. Since no one has studied this, we don’t know.
3 Likes
Maybe Dr. Salmon has information about this from the marmoset studies.
4 Likes
Sirt6
#18
@LaraPo has the thymus size been checked in your case?
Many thanks!
I could buy an ultrasound device and check my thymus on a regular basis.
Another approach would be to cycle rapamycin and HGH. But we don’t know, if this could cancel any benefits. We would have to define and check bioparameters.
1 Like
jakexb
#19
Is thymus always correlated with thymus function? Honestly don’t know, just curious if there is anything in the thymus similar to some other organs where they enlarge in an attempt to compensate for not working as well with age.
LaraPo
#20
I have never checked on my thymus. None of my CPs ever mentioned that. I don’t think that insurance would cover it either. Will ask though at my next visit.
2 Likes
Jjazz
#21
Thymus MRI is going to expensive and, as you suggested, not covered by insurance. A blood test that measures circulating naive T cells would be a more practical readout that would reflect thymus function. Something like this:
https://www.mayocliniclabs.com/test-catalog/overview/89319
2 Likes