Related paper: Reduced Prevalence of Parkinson’s Disease in Patients Prescribed Calcineurin Inhibitors 2024
We analyzed electronic health records (EHRs) from patients prescribed the brain penetrant CNI tacrolimus (TAC), the peripherally restricted CNI cyclosporine (CySp), or the non-CNI sirolimus (SIR).
Our results suggest CNIs, especially those acting within the brain, may prevent PD. The reduced prevalence of PD in patients prescribed TAC, compared to patients prescribed SIR, suggests that mechanisms of calcineurin inhibition— other than immunosuppression, which is common to both drugs— are driving the reduction. Therefore, CNIs may provide a promising therapeutic approach for PD.
mTORC1-Dependent Protein and Parkinson’s Disease: A Mendelian Randomization Study 2023
The mTOR pathway is crucial in controlling the growth, differentiation, and survival of neurons, and its pharmacological targeting has promising potential as a treatment for Parkinson’s disease. However, the function of mTORC1 downstream proteins, such as RPS6K, EIF4EBP, EIF-4E, EIF-4G, and EIF4A, in PD development remains unclear. […] The genetic proxy EIF4EBP was found to be inversely related to PD risk (OR = 0.79, 95% CI = 0.67–0.92, p = 0.003), with the results from WM, MR-PRESSO, and MR-RAPS being consistent. […] Our unbiased MR study highlights the protective role of serum EIF4EBP levels in PD, suggesting that the pharmacological activation of EIF4EBP activity could be a promising treatment option for PD.”
Also, the debate about statins and PD continues (@A_User FYI, I couldn’t add a reply so I’m editing this previous message), and it’s not getting clearer: Statin use and risk of Parkinson’s disease among older adults in Japan: a nested case-control study using the longevity improvement and fair evidence study 2024
The inverse association between statin use and Parkinson’s disease risk was significant after adjusting for multiple variables (odds ratio: 0.61; 95% confidence interval: 0.56–0.66). Compared with non-users, the dose analysis revealed varying odds ratios: 1.30 (1.12–1.52) for 1–30 total standard daily doses, 0.77 (0.64–0.92) for 31-90 total standard daily doses, 0.62 (0.52–0.75) for 91–180 total standard daily doses, and 0.30 (0.25–0.35) for >180 total standard daily doses. Statin use among older Japanese adults was associated with a decreased risk of Parkinson’s disease. Notably, lower cumulative statin doses were associated with an elevated risk of Parkinson’s disease, whereas higher cumulative doses exhibited protective effects against Parkinson’s disease development.
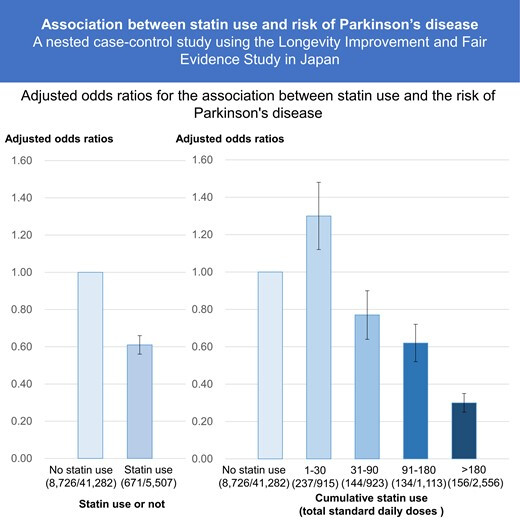
People using low-dose statins have a higher PD risk than statin non-users but people using high-dose statins have a lower risk than non-users? I’m struggling to understand how this could make sense and not be the result of some confounding factors. They don’t address this problem seriously in the paper. But they note later:
Finally, our study was not adjusted for potential confounding factors. The absence of data on BMI, smoking, and alcohol consumption (potential confounders) may have led to an overestimation of the protective effects of statins in our study. We used the E-value to estimate the influence of the unmeasured confounders. If the unmeasured confounders were associated with both statin use and PD risk with an OR of 2.66 or higher, the result was explained by unmeasured confounders.29 Future studies should focus on comprehensive adjustments for these confounding variables to provide a more accurate assessment of the effects of statins.
As we know that low LDL is associated with PD and that this association might be causal, and that high BMI seems protective of PD, this is probably the cofounder: people on high-dose statin are overweight and had high LDL most of their life, which protected them from PD. The negative effect seen at low-dose might be for people with slightly elevated LDL, so who are not protected that much from PD, but who get the negative side effects of statins (glucose dysregulation and gut dysbiosis, that are detrimental for PD).
So I would conclude the exact opposite as the authors based on this paper: it probably confirms that non-statin lipid-lowering therapies should be used first in people with PD or at high risk of (family history, environment, genes, etc.).