Flow published their results in Nature Medicine in October: Home-based transcranial direct current stimulation treatment for major depressive disorder: a fully remote phase 2 randomized sham-controlled trial 2024
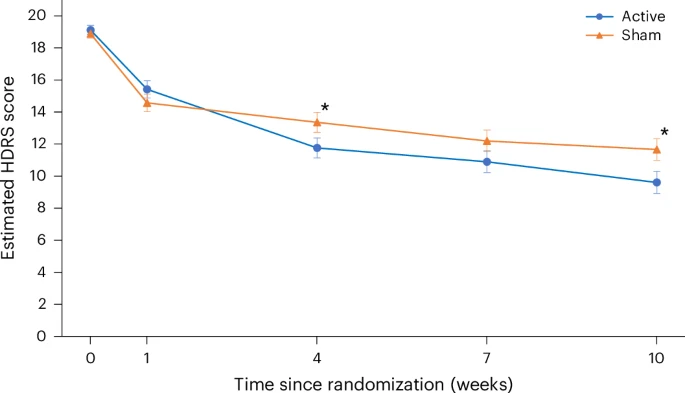
Transcranial direct current stimulation (tDCS) has been proposed as a new treatment in major depressive disorder (MDD). This is a fully remote, multisite, double-blind, placebo-controlled, randomized superiority trial of 10-week home-based tDCS in MDD. Participants were 18 years or older, with MDD in current depressive episode of at least moderate severity as measured using the Hamilton Depression Rating Scale (mean = 19.07 ± 2.73). A total of 174 participants (120 women, 54 men) were randomized to active (n = 87, mean age = 37.09 ± 11.14 years) or sham (n = 87, mean age = 38.32 ± 10.92 years) treatment. tDCS consisted of five sessions per week for 3 weeks then three sessions per week for 7 weeks in a 10-week trial, followed by a 10-week open-label phase. Each session lasted 30 min; the anode was placed over the left dorsolateral prefrontal cortex and the cathode over the right dorsolateral prefrontal cortex (active tDCS 2 mA and sham tDCS 0 mA, with brief ramp up and down to mimic active stimulation). As the primary outcome, depressive symptoms showed significant improvement when measured using the Hamilton Depression Rating Scale: active 9.41 ± 6.25 point improvement (10-week mean = 9.58 ± 6.02) and sham 7.14 ± 6.10 point improvement (10-week mean = 11.66 ± 5.96) (95% confidence interval = 0.51–4.01, P = 0.012). There were no differences in discontinuation rates. In summary, a 10-week home-based tDCS treatment with remote supervision in MDD showed high efficacy, acceptability and safety. ClinicalTrials.gov registration: NCT05202119
Inclusion criteria included being treatment-free or taking stable antidepressant medication or undergoing psychotherapy for at least 6 weeks before enrollment.
The composition of the participant cohort was as follows: treatment-free = 57 (32.8%); taking antidepressant medication = 109 (62.6%); undergoing psychotherapy = 26 (14.9%); taking medication and undergoing psychotherapy = 18 (10.3%) participants.
Based on the HDRS ratings, the active tDCS treatment arm was associated with a significantly greater clinical response of 58.3% compared to the sham treatment arm (37.8%; P = 0.017) (post hoc odds ratio (OR) = 2.31 (lower bound = 1.17, upper bound = 4.55); the active treatment arm was associated with a significantly greater remission rate of 44.9% relative to the sham treatment arm (21.8%; P = 0.004) (post hoc OR = 2.93, lower bound = 1.41, upper bound = 6.09).
There were no significant differences in quality of life between treatment arms as measured by EQ-5D-3L
Regarding anxiety symptoms, there were no significant differences between an active mean Hamilton Anxiety Rating Scale (HAM-A) score improvement of 6.62 (s.d. = 6.09) (mean = 8.24 (s.d. = 5.65)), compared to a sham improvement of 4.88 (s.d. = 5.88) (mean = 9.29 (s.d. = 4.90)) (P = 0.08).
In the neuropsychological assessments, there were no significant differences in Rey Auditory Verbal Learning Test (RAVLT) total learning or Symbol Digit Modalities Test (SDMT) between treatment arms (Supplementary Table 14).
Before unblinding at week 10 (end of trial), participants were asked to guess whether they thought they were receiving the active or sham tDCS device and their level of certainty, rating from ‘1’ for ‘very uncertain’ to ‘5’ for to ‘very certain’. A guess of active tDCS was made by 77.6% in the active treatment arm and 59.3% in the sham treatment arm; the difference was significant (P = 0.01).
Furthermore, when using the sham device, there was brief stimulation at the start and at the end of each session to mimic active tDCS sensations to aid in blinding and to balance potential nocebo effects across groups
As the authors, other trials previously found no benefits for tDCS, but they last 6 weeks instead of 10.
Results among antidepressant-free patients:
“clinical response defined as a minimum of 50% reduction from baseline in HDRS, MADRS and MADRS-s at week 10; clinical remission defined as an HDRS score of 7 or less, MADRS score of 10 or less and MADRS-s score of 12 or less”
ChatGPT says that typically the HDRS response and remissions ORs are around 1.5–2 in drug-free patients. Here we have 6–9! So if correct, it means that in drug-free patients, tDCS is very good. ChatGPT notes: “The categorical measures (response/remission) look extremely impressive (massive ORs), even though the overall mean score reduction is not dramatically larger than what is usually seen with standard antidepressants. This pattern suggests that the treatment might be especially effective in nudging patients over important clinical thresholds rather than uniformly lowering symptom severity by a large margin across the entire sample.”
The reviews are also impressive: Flow Neuroscience Reviews | Read Customer Service Reviews of flowneuroscience.com
I’m more and more intrigued…
This meta-analysis found: Transcranial Magnetic Stimulation and Transcranial Direct Current Stimulation Across Mental Disorders A Systematic Review and Dose-Response Meta-Analysis 2024: “The study findings suggest that NIBS yields specific outcomes based on dose parameters across various mental disorders and brain regions. Clinicians should consider these dose parameters when prescribing NIBS. Additional research is needed to prospectively validate the findings in randomized, sham-controlled trials and explore how other parameters contribute to the observed dose-response association.”