Association of Plasma Omega-3 Levels With Incident Heart Failure and Related Mortalities 2024
In participants without HF at baseline (n=271,794), a generally linear inverse association was observed between omega-3 levels and incident HF during a median follow-up of 13.7 years. The risk was 21% lower in the highest quintile of omega-3 compared with the lowest quintile (hazard ratio, 0.79; 95% CI, 0.74 to 0.84; P<.001) in multivariable models.
In parallel models in participants with prevalent HF (n=1239), risk for all-cause and CV mortality were both reduced by approximately 50% comparing top to bottom omega-3 quintiles (hazard ratio, 0.53; 95% CI, 0.33 to 0.86; and hazard ratio, 0.50; 95% CI, 0.31 to 0.79, respectively; both P<.01).
The two interesting charts:
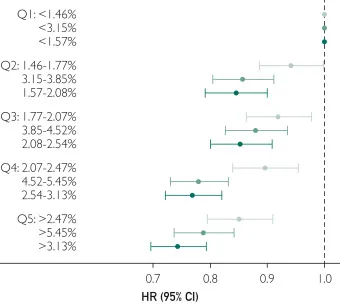
Figure 1. Forest plot illustrating the association between omega-3 plasma levels and the incidence of heart failure (HF). As omega-3 plasma levels increase, there is a concurrent decrease in the risk (ie, lower hazard ratio [HR]) of HF. Hazard ratios associated with docosahexaenoic acid (DHA) are shown in light gray, with total omega-3 in light green and non-DHA in dark green. Adjusted for age, sex, race/ethnicity, history of coronary heart disease, low-density lipoprotein to high-density lipoprotein cholesterol ratio, cholesterol medication, hypertension, diabetes mellitus, smoking status, alcohol consumption, level of education, exercise, body mass index, and waist circumference. Q, quintile.
Figure 2. Forest plots depicting the relationship between omega-3 plasma levels and all-cause mortality (A) and cardiovascular mortality (B). As omega-3 plasma levels increase, there is a corresponding decrease in the risk of all-cause mortality (A) and cardiovascular mortality (B). Hazard ratios associated with DHA are shown in light gray, with total omega-3 in dark green, and non-DHA in black. Adjusted for age, sex, race/ethnicity, history of coronary heart disease, low-density lipoprotein to high-density lipoprotein cholesterol ratio, cholesterol medication, hypertension, diabetes mellitus, smoking status, alcohol consumption, level of education, exercise, body mass index, and waist circumference. Q, quintile.
So to be in the top quintile, you need:
- Serum DHA: >2.47%
- Serum total omega 3: >5.45% (
 This is NOT the omega 3 index as the omega 3 index looks at RBC and not serum
This is NOT the omega 3 index as the omega 3 index looks at RBC and not serum  )
)
- Serum non-DHA: >3.13%
Regarding non-DHA, they note:
In our study, when comparing the plasma levels of DHA, non-DHA, and total omega-3 as exposure markers, associations with risk for HF were somewhat stronger for non-DHA than DHA. For the mortality outcomes, differences in HRs were reasonably similar with all 3 metrics. An interlaboratory experiment (see Patients and Methods) confirmed that the strongest predictor of non-DHA omega-3 levels was EPA (R2=46.8%), while neither alpha-linolenic acid nor docosapentaenoic acid were statistically significant predictors (P>.05 in both cases) of the non-DHA omega-3 level. Thus, it appears (although only inferred) that a higher EPA level may have been more directly linked to the lower incidence of HF than a higher DHA level. This finding aligns with the results from the Multi-Ethnic Study of Atherosclerosis (MESA), which reported that higher plasma EPA levels were significantly associated with reduced risk for HF after 13 years of follow-up.