30% to 40% CR is really hard… you’re always hungry, you lose a ton of fat so you’re always cold, your sex drive drops to Zero… its not fun. A very small percent of people will stick with CR for a year or more. 10% probably not a big issue. At that level you’re not (in most people’s minds) not really doing true CR.
SGLT2i is not at all like CR in terms of the user experience. You get to eat your regular food (hopefully healthy)… you feel full, you aren’t hungry all the time, but you do lose weight (at least for the first month or so). The only really side effect that impacts everyone is increased urination… so you’ll have to decide for yourself whether the benefits for you are worth that “cost”. I think anyone above about a BMI of 20 will see weight loss with SGLT2. I’m well below BMI of 30 and lost about 10 to 15lbs. I had slightly better weight loss with Metformin though. I’d like to get my body fat down to 10%.
And comparing CR vs. SGLT21 vs. Rapamycin. Rapamycin I get no side effects (noticeable ones at least) at all. No weight loss either (though over the long term, perhaps loss of visceral fat… I’m going to start doing DEXA scans going forward to monitor this).
5 Likes
Neo
#543
There seems to me to be an ever growing body of evidence that optimal gut biome is part of both health and life extension.
So I think you may want to weight that in any decision you make for longevity - including that there seems to be real evidence for that that happens with SGLT1i
That might be a good hypothesis to explore and it could be the case.
But I think it’s important to note that believing in that more than at a theory or hypothesis levels is just a leap of faith.
adssx
#544
I asked them a few weeks ago to look at SGLT2 in longitudinal data. Their primary interest lies in neurodegenerative diseases (mainly AD, PD, DLB, ALS, and MS) so they need at least 5y of history and they told me that they didn’t have enough people on SGLT2 in their cohorts as this class of drugs is quite recent. But they should have access to larger databases soon. I’ll ask again. Smart to look at the UK biobank. Do you have in mind something like the Levine age before/after SGLT2 prescription? (compared to before/after metformin, GLP-1RA, etc.)
3 Likes
Neo
#545
That great 
Yes, something like that - you could run the whole suite of blood based are clocks. Perhaps one of the many epigentics groups or labs would want to run their biological are clocks on it too.
Re brain - this group has a brain biological aging clock
1 Like
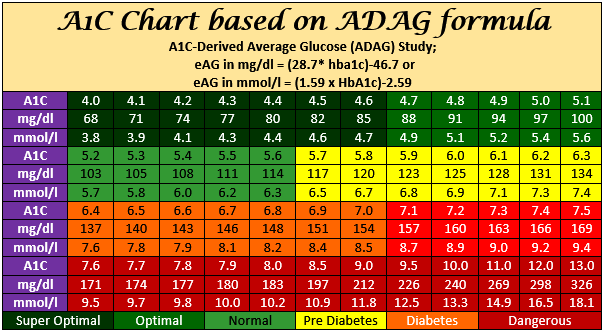
Since empagliflozin drops HBA1C by about 1.5, and my HBA1C was 5.7 (probably lower now), is it possible it would drop me below 4.0? Or is there a baseline amount it won’t take you below?
1 Like
It’s almost impossible to drop below 4%. It would means you’re strongly hypoglycemic all the time (or maybe if you give blood a lot!). Beetwen 4.5% and 5.5% is really good.
7 Likes
adssx
#548
That’s probably among diabetic people. Among non diabetic, I’d assume the reduction is way smaller.
3 Likes
True, but do we have any data to help quantify a drop in pre diabetic individuals?
1 Like
adssx
#550
Empagliflozin treatment effects across categories of baseline HbA1c, body weight and blood pressure as an add‐on to metformin in patients with type 2 diabetes 2021:
Hb A1C below 8.5%: –0.5 points of HbA1C only.
So I’d guess that if you’re pre-diabetic, the impact on HbA1C will be almost 0. SGLT2 inhibitors reduce big spikes mostly.
4 Likes
Neo
#552
Found the theory that SGLTi generates a kind of marginal hibernation in us with lower metabolic rate, and hunker down pathways activated, very interesting (believe that hibernation animals tend to live longer than their paired animal family brethren)
For anyone who just wants to watch the video for a few min the section around 7:00 (sound get better soon) and around 44:25
This is the main paper on that, in a good journal
https://www.nature.com/articles/s41581-020-00350
3 Likes
adssx
#553
Thanks a lot for sharing this. Here’s the correct link btw: Organ protection by SGLT2 inhibitors: role of metabolic energy and water conservation | Nature Reviews Nephrology
It’s indeed a good journal, and it’s actually authored by Boehringer Ingelheim Japan. Boehringer Ingelheim is the manufacturer of empagliflozin.
3 Likes
adssx
#554
Effect of pharmacological selectivity of SGLT2 inhibitors on cardiovascular outcomes in patients with type 2 diabetes: a meta-analysis 2024
Pharmacological selectivity of these agents to SGLT2 over SGLT1 is highly variant, with unknown clinical relevance. Genetically reduced SGLT1—but not SGLT2—activity correlates with lower risk of heart failure and mortality, therefore additional non-selective SGLT1 inhibition might be beneficial. […] However, lower SGLT2 selectivity correlated with significantly lower risk of stroke (pseudo-R2 = 78%; p = 0.011). Indeed, dual SGLT1/2 inhibitors significantly reduced the risk of stroke (hazard ratio [HR], 0.78; 95% confidence interval [CI], 0.64–0.94), unlike selective agents (p for interaction = 0.018). […] However, hypotension occurred more often with non-selective SGLT2 inhibitors (odds ratio [OR], 1.87; 95% CI, 1.20–2.92) compared with selective agents (p for interaction = 0.044). In conclusion, dual SGLT1/2 inhibition reduces stroke in high-risk T2DM patients but has limited additional effect on other clinical outcomes.
@Neo this paper is for you! It’s only a meta analysis of 6 trials but still interesting trend/signal.
They also write: “Pharmacological selectivity of SGLT2 inhibitors did not affect all-cause death (pseudo-R2 = 10%; p = 0.25) (Fig. 1). Indeed, neither non-selective (HR, 0.90; 95% CI, 0.80–1.01) nor highly selective (HR, 0.84; 95% CI, 0.69–1.03) SGLT2 inhibitors reduced all-cause death as compared with placebo (p = 0.65)” but I mean it’s very close to statistical significance. When I see the huge reduction in hospitalisation (Fig. 2) I assume that with more and longer trials we would see a significant 10-15% reduction in all-cause mortality.
Anyway, another paper that makes me bullish on sotagliflozin…
3 Likes
adssx
#555
Actually, looking at Figure 1, sota is not that exciting, especially in all-cause deaths. Empa and cana are the leaders. But again, only 6 trials… (and it’s among T2D)
4 Likes
SGLT2 Inhibitors: A Potential Game-Changer in Kidney Stone Prevention for Diabetic Patients
In recent years, the incidence of kidney stones has been on the rise, both in the United States and globally. This increase is particularly notable among individuals with type 2 diabetes, who are at a heightened risk of developing these painful formations. However, new research suggests that a particular type of diabetes medication, Sodium Glucose Cotransporter 2 (SGLT2) inhibitors, may hold the key to reducing this risk.
4 Likes
adssx
#557
More and more papers on SGLTi and liver health: Empagliflozin Reduces Liver Fat in Individuals With and Without Diabetes 2024
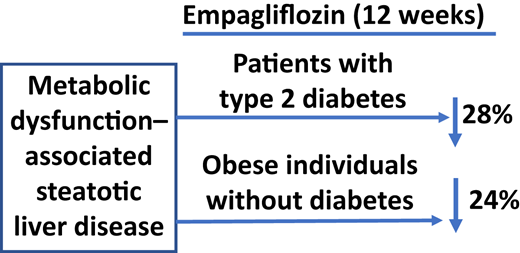
Empagliflozin is effective in reducing liver fat content in individuals with and without T2D. The decrease in liver fat content is independent of the decrease in plasma glucose concentration and is strongly related to the decrease in body weight and improvement in insulin sensitivity.
There are dozens of ongoing trials of SLGTi (mostly dapagliflozin and empagliflozin) for liver diseases such as NASH (Non-Alcoholic Steatohepatitis), NAFLD (Non-Alcoholic Fatty Liver Disease), and liver cirrhosis/fibrosis. Most of them are run by academia, charities or public bodies, but two are conducted by big pharma:
So I guess that after T1D, T2D, CKD, and HF, SGLTi will soon be expanded to all liver indications. Quite an impressive class of drugs if in addition to the heart and kidney, it can protect the liver.
Let’s assume (for the sake of argument) that SGLTi can indeed significantly slow down organ aging in the heart, kidney, and liver. Which organs would be left to protect? The brain, lungs, and gut mostly?
8 Likes
I started taking Jardiance based on all the enthusiasm here, but further research gives me pause. This write-up says it’s not recommended if egfr is below 45.
Sodium-Glucose Cotransporter-2 (SGLT2) Inhibitors: A Clinician’s Guide - PMC
These thresholds change depending on which document your read. I found one (which I can’t find now) that said if egfr is between 45-60, cana and empa (iirc) should have dosage ‘adjusted’, whereas dapa should not be initiated with egfr below 60. 60 is not all that low. My creatinine of 1.50 mg/dl produces an egfr around 50. (OTOH, cystatin-C says egfr of 86. Go figure.)
Furthermore, here’s another one which mentions a sharp decline in egfr after slgt2 iinitiation:
Sodium-Glucose Transport Protein 2 (SGLT2) Inhibitors - StatPearls - NCBI Bookshelf
A 30% decline in egfr would seem rather disturbing. Has any DIYer out there looked at their egfr before and after slgt2 initiation?
5 Likes
adssx
#559
I feel like the thresholds are for T2D patients, due to the lack of glucose-lowering effect if eGFR < 45: “All SGLT2i have limited glycaemic effect when eGFR<45. Local consensus is to discontinue any SGLT2i if eGFR <45, where used solely for glycaemic effect in T2DM, although the licenses may be lower.” (source, see also this)
But SGLT2i are also approved for CKD, and they’re used continuously until end-stage kidney disease (ESKD), meaning eGFR < 15. From the above source:
Indeed, SGLTi are highly beneficial to people with low eGFR: Characteristics predicting the efficacy of SGLT-2 inhibitors versus GLP-1 receptor agonists on major adverse cardiovascular events in type 2 diabetes mellitus: a meta-analysis study 2023
Similarly, in the meta-analysis, SGLT-2i therapy tended to be more effective in reducing 3P-MACE in people with eGFR < 60 ml/min/1.73 m2 than in those with normal renal function (ARR − 0.90 [–1.44 to − 0.37] vs. − 0.17 [–0.34 to − 0.01] events/100 person-years).
There’s indeed a dip at initiation but then it stays constant instead of declining in CKD: The potential for improving cardio-renal outcomes by sodium-glucose co-transporter-2 inhibition in people with chronic kidney disease: A rationale for the EMPA-KIDNEY study 2018
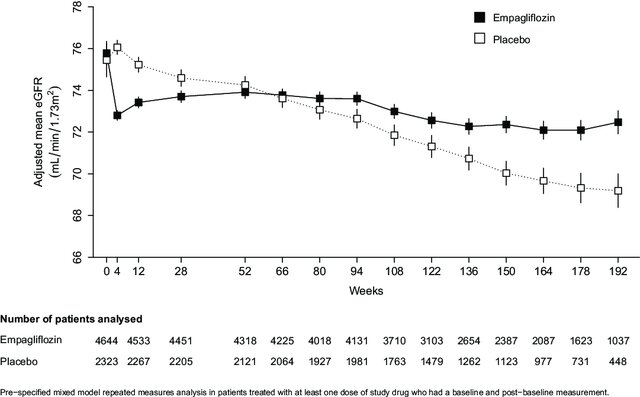
I don’t know anything about eGFR tbh but this article says: “Dapagliflozin was associated with an acute dip in eGFR on initiation suggestive of a beneficial hemodynamic effect” (Prescribing SGLT2 Inhibitors in Patients With CKD: Expanding Indications and Practical Considerations 2022)
3 Likes
adssx
#560
This is the topic of this article, with a wonderful title: eGFR Decline after SGLT2 Inhibitor Initiation: The Tortoise and the Hare Reimagined 2021
Furthermore, clinicians need to know what to expect when prescribing these agents, including an early decline in eGFR after initiation.
These drugs inhibit sodium and glucose reabsorption in the proximal tubule, leading to increased sodium and chloride delivery to the macula densa. This results in afferent arteriolar vasoconstriction secondary to adenosine-mediated myogenic activation, leading to a reduction in the intraglomerular pressure and the GFR. Therefore, it is not surprising that the major SGLT2i outcome trials have reported an early decline in eGFR (around 3–6 ml/min per 1.73 m2) shortly after initiating these drugs compared with placebo controls. These early declines or dips were typically observed at 2–4 weeks after initiation of the SGLT2i, with subsequent partial recovery of the eGFR curve by week 12, and, ultimately, followed by an attenuation of the slope of eGFR decline compared with placebo controls after 52 weeks
How can we reconcile early dips in eGFR with long-term nephroprotection? […] Hyperfiltration is frequently seen in diabetic kidney disease, and often precedes the development of overt albuminuria. Therefore, mitigation of pathologic hyperfiltration may be therapeutic, even at the cost of an acute eGFR decline. These data lend credence to the somewhat counterintuitive notion that agents that cause initial decrements in eGFR have long-term therapeutic benefits for patients with proteinuric kidney disease.
In clinical practice, SGLT2i prescribers, who will frequently be non-nephrologists, may be uncomfortable watching the serum creatinine rise in response to initiating these agents. This anxiety may have been inadvertently heightened by a sensitivity to “causing” AKI. […] Serum creatinine rises of similar magnitude are unlikely to have the same significance in an otherwise stable outpatient who initiated an SGLT2i.
The seemingly paradoxic coexistence of eGFR declines and long-term clinical benefits has precedent in nephrology. This phenomenon is well described with renin-angiotensin-aldosterone system inhibitors.
How does a clinician respond when faced with an abrupt rise in serum creatinine after initiation of an SGLT2i? Although the data cited above would suggest that most such dips are merely expected hemodynamic changes of limited clinical relevance, it is essential to recognize that, in some cases, this may signal systemic illness (i.e., infection, occult bleeding) with bona fide kidney injury. Traditionally, a 30% increase in serum creatinine has prompted clinicians to re-evaluate renin-angiotensin-aldosterone system blockade, and we believe the same approach may be reasonable for SGLT2is.
See also Table 2. Characteristics of patients at baseline in kidney outcome trials. in the just published SGLT2 Inhibitors to Slow Chronic Kidney Disease Progression: A Review. They started treating people with mean eGFR between 30 and 60:
And they of course had crazy good outcomes:
As the authors note:
The tubuloglomerular feedback theory also explains the initial eGFR “dip” commonly seen with initiation of SGLT2 inhibitors. In CREDENCE, most patients had an eGFR measured at baseline and week 3, with 45% of the canagliflozin group experiencing an acute eGFR drop >10%, with an acute drop in eGFR >30% being a rare event (4.2% of canagliflozin group). For all groups with an acute eGFR drop (0%–10%, 10%–30%, and >30%), the annual decline in eGFR was lower in the canagliflozin group. Similarly, in DAPA-CKD, 49.4% of the dapagliflozin group experienced an acute eGFR drop >10% at week 2, with only 4.7% having an eGFR reduction >30%. For all 3 groups, the acute eGFR drop did not change long-term clinical outcomes. Based on these data, an eGFR decline of 10% to 30% can be expected in about one half of patients who initiate an SGLT2 inhibitor and has not been shown to adversely affect long-term benefits.
5 Likes
I wouldn’t rely on a creatinine base eGFR. I’d get a cystatin-c test for a more accurate assessment of kidney function.
4 Likes